Cusabio Human Recombinants
Recombinant Human Heat shock protein HSP 90-alpha (HSP90AA1), partial | CSB-YP010802HU1
- SKU:
- CSB-YP010802HU1
- Availability:
- 25 - 35 Working Days
Description
Recombinant Human Heat shock protein HSP 90-alpha (HSP90AA1), partial | CSB-YP010802HU1 | Cusabio
Alternative Name(s): Heat shock 86KDA ;HSP 86 ;HSP86Lipopolysaccharide-associated protein 2 ;LAP-2 ;LPS-associated protein 2Renal carcinoma antigen NY-REN-38
Gene Names: HSP90AA1
Research Areas: Neuroscience
Organism: Homo sapiens (Human)
AA Sequence: DQPMEEEEVETFAFQAEIAQLMSLIINTFYSNKEIFLRELISNSSDALDKIRYESLTDPSKLDSGKELHINLIPNKQDRTLTIVDTGIGMTKADLINNLGTIAKSGTKAFMEALQAGADISMIGQFGVGFYSAYLVAEKVTVITKHNDDEQYAWESSAGGSFTVRTDTGEPMGRGTKVILHLKEDQTEYLEERRIKEIVKKHSQFIGYPITLFVEKERDKEVSD
Source: Yeast
Tag Info: N-terminal 6xHis-tagged
Expression Region: 9-232aa
Sequence Info: Partial
MW: 27.2 kDa
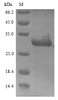
Purity: Greater than 90% as determined by SDS-PAGE.
Relevance: Molecular chaperone that promotes the maturation, structural maintenance and proper regulation of specific target proteins involved for instance in cell cycle control and signal transduction. Undergoes a functional cycle that is linked to its ATPase activity. This cycle probably induces conformational changes in the client proteins, thereby causing their activation. Interacts dynamically with various co-chaperones that modulate its substrate recognition, ATPase cycle and chaperone function. Binds bacterial lipopolysaccharide (LPS) et mediates LPS-induced inflammatory response, including TNF secretion by monocytes.
Reference: Nucleotide sequence of a full-length cDNA for 90KDA heat-shock protein from human peripheral blood lymphocytes.Soeda E., Yokoyama K., Yamazaki M., Akaogi K., Miwa T., Imai T.Nucleic Acids Res. 17:7108-7108(1989)
Storage: The shelf life is related to many factors, storage state, buffer ingredients, storage temperature and the stability of the protein itself. Generally, the shelf life of liquid form is 6 months at -20?/-80?. The shelf life of lyophilized form is 12 months at -20?/-80?.
Notes: Repeated freezing and thawing is not recommended. Store working aliquots at 4? for up to one week.
Function: Molecular chaperone that promotes the maturation, structural maintenance and proper regulation of specific target proteins involved for instance in cell cycle control and signal transduction. Undergoes a functional cycle that is linked to its ATPase activity which is essential for its chaperone activity. This cycle probably induces conformational changes in the client proteins, thereby causing their activation. Interacts dynamically with various co-chaperones that modulate its substrate recognition, ATPase cycle and chaperone function
Involvement in disease:
Subcellular Location: Nucleus, Cytoplasm, Melanosome, Cell membrane
Protein Families: Heat shock protein 90 family
Tissue Specificity:
Paythway: Estrogensignalingpathway
Form: Liquid or Lyophilized powder
Buffer: If the delivery form is liquid, the default storage buffer is Tris/PBS-based buffer, 5%-50% glycerol. If the delivery form is lyophilized powder, the buffer before lyophilization is Tris/PBS-based buffer, 6% Trehalose, pH 8.0.
Reconstitution: We recommend that this vial be briefly centrifuged prior to opening to bring the contents to the bottom. Please reconstitute protein in deionized sterile water to a concentration of 0.1-1.0 mg/mL.We recommend to add 5-50% of glycerol (final concentration) and aliquot for long-term storage at -20?/-80?. Our default final concentration of glycerol is 50%. Customers could use it as reference.
Uniprot ID: P07900
HGNC Database Link: HGNC
UniGene Database Link: UniGene
KEGG Database Link: KEGG
STRING Database Link: STRING
OMIM Database Link: OMIM